Urology treats urinary tract diseases in all genders and male reproductive issues, covering the kidneys, bladder, prostate, urethra, from infections to complex cancers.
Send us all your questions or requests, and our expert team will assist you.
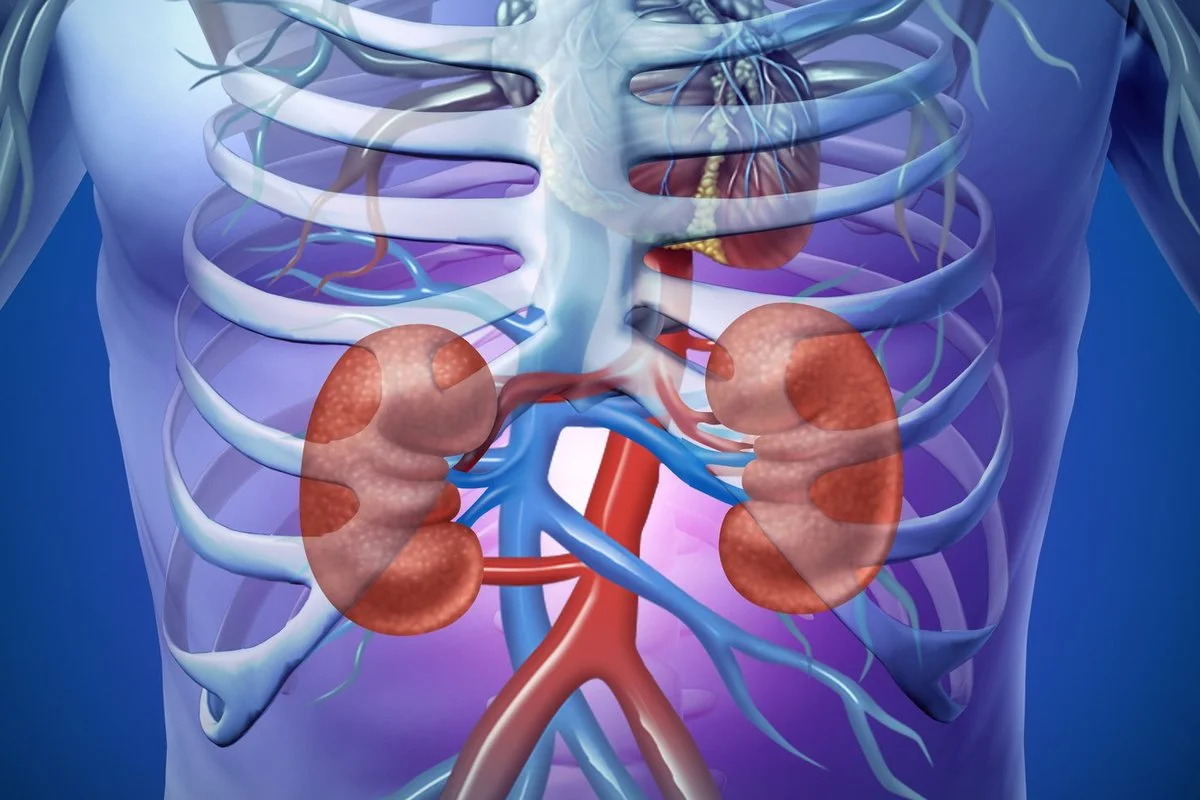
Interventional Nephrology represents a specialized discipline at the intersection of nephrology, interventional radiology, and vascular surgery. It is defined by the application of minimally invasive, image-guided procedures to manage the vascular access needs of patients with End-Stage Renal Disease (ESRD) and to treat structural abnormalities of the renal system. In the modern era of regenerative medicine, this field has expanded beyond mechanical troubleshooting to encompass the biological optimization of the vascular endothelium and the preservation of the dialysis circuit. The core objective is to maintain a functional lifeline for hemodialysis while minimizing the systemic burden of repeated interventions.
The physiological foundation of Interventional Nephrology lies in the management of the Arteriovenous Fistula (AVF) and the Arteriovenous Graft (AVG). These vascular conduits are engineered to sustain high-volume blood flow, often exceeding 600 mL/min, which creates a unique hemodynamic environment. This high shear stress triggers profound cellular responses within the vessel wall, including endothelial activation and smooth muscle proliferation. Interventional Nephrology seeks to modulate these responses, preventing the pathological remodeling that leads to stenosis and thrombosis. The discipline also includes the placement and management of peritoneal dialysis catheters, renal biopsy, and the treatment of renovascular hypertension through renal artery angioplasty and stenting.
From a cellular perspective, the field is increasingly focused on the biology of the vessel wall. The maturation of an AV fistula requires a coordinated process of outward remodeling, driven by the degradation of the extracellular matrix and the realignment of endothelial cells. Failure of maturation, a common clinical challenge, is often due to aggressive neointimal hyperplasia, a proliferative response driven by inflammatory cytokines and growth factors. Interventional nephrologists now utilize drug-coated balloons and stents that elute anti-proliferative agents, directly targeting this cellular pathology to extend the patency of the access.
The creation of an arteriovenous access initiates a molecular shock to the venous system. The sudden exposure of the vein to arterial pressure and oxygenation alters the expression of genes regulating vascular tone and inflammation. Hypoxia-Inducible Factor 1-alpha (HIF-1alpha) is upregulated in areas of disturbed flow, driving the expression of Vascular Endothelial Growth Factor (VEGF). While VEGF promotes necessary angiogenesis, its overexpression can lead to leaky, fragile microvessels within the vessel wall, contributing to plaque hemorrhage and restenosis.
Transforming Growth Factor-beta (TGF-beta) plays a dual role in this environment. It is essential for the synthesis of collagen and the stabilization of the fistula, but its dysregulation leads to fibrosis and stiffening of the vein. This fibrotic remodeling reduces the compliance of the vessel, making it prone to stenosis. Interventional strategies are evolving to include the local delivery of agents that modulate TGF-beta signaling, aiming to balance necessary structural reinforcement with the prevention of pathological fibrosis. The molecular landscape also involves the upregulation of Matrix Metalloproteinases (MMPs), which degrade the elastic lamina, facilitating the migration of smooth muscle cells into the intima, the primary mechanism of neointimal hyperplasia.
Global trends in Interventional Nephrology are characterized by a shift towards “EndoAVF” creation and bio-engineered vascular grafts. EndoAVF technology utilizes catheter-based systems to create an arteriovenous fistula between the proximal radial artery and the perforating vein using thermal or radiofrequency energy. This approach avoids open surgical dissection, preserving the vasa vasorum and the perivascular nervous plexus. The result is a fistula with less surgical trauma, reduced inflammation, and potentially superior long-term patency.
Biotechnology is also driving the development of tissue-engineered vascular grafts. These bio-intelligent conduits are created by seeding biodegradable scaffolds with the patient’s own endothelial and smooth muscle cells. As the scaffold degrades, it is replaced by native extracellular matrix, resulting in a living, self-repairing vessel that resists infection and thrombosis. This represents a paradigm shift from the use of inert synthetic materials like PTFE to regenerative solutions that integrate seamlessly with the patient’s physiology.
The extracellular matrix (ECM) of the vascular wall dictates the mechanical properties of the dialysis access. In a healthy fistula, the matrix must be dynamic, allowing for significant dilation. In pathological states, the accumulation of advanced glycation end-products (AGEs), particularly in diabetic patients, leads to cross-linking of collagen fibers. This “glycation” stiffens the matrix, impairing the vessel’s ability to dilate (failure to mature) and increasing the risk of rupture during cannulation.
Regenerative interventions aim to rejuvenate the ECM. Research into the use of elastase inhibitors seeks to prevent the fragmentation of elastin fibers, preserving the recoil capacity of the vessel. Furthermore, the application of paclitaxel or sirolimus-coated balloons during angioplasty inhibits the secretion of excess collagen by fibroblasts, maintaining the luminal diameter. The integrity of the basement membrane is also critical; it serves as the anchor for the endothelium. Disruption of this membrane during aggressive angioplasty can lead to endothelial detachment and acute thrombosis, highlighting the need for gentle, biologically respectful interventions.
The application of energy is central to many interventional nephrology procedures. Radiofrequency ablation is used in the creation of EndoAVFs and in renal sympathetic denervation for resistant hypertension. The energy dynamics must be precisely controlled to induce thermal fusion or nerve modulation without causing transmural necrosis. In the context of thrombectomy, mechanical and rheolytic devices utilize fluid dynamics and kinetic energy to fragment and aspirate clots.
The interaction between these energy sources and the blood components is critical. Excessive shear forces or thermal energy can induce hemolysis, releasing free hemoglobin which is toxic to the renal tubules. Modern devices are designed to minimize this hematological trauma. The use of intravascular ultrasound (IVUS) employs sound waves to characterize the vessel wall composition, distinguishing between soft thrombus, fibrous plaque, and calcium. This “energy-based visualization” guides the selection of the appropriate therapy, ensuring that the intervention targets the specific pathology without damaging the healthy vessel architecture.

Send us all your questions or requests, and our expert team will assist you.
The “Rule of 6s” is a clinical guideline used to determine if an arteriovenous fistula is mature enough for dialysis. It states that the fistula should be at least 6 mm in diameter, less than 6 mm deep under the skin, and have a blood flow of at least 600 mL/min. Meeting these criteria ensures the access can support the high flow rates required for effective hemodialysis.
A drug-coated balloon is an angioplasty balloon covered with a medication, typically paclitaxel or sirolimus. When the balloon is inflated to open a narrowed vessel, it transfers the drug into the vessel wall. These drugs inhibit the proliferation of smooth muscle cells and scar tissue formation (neointimal hyperplasia), significantly reducing the likelihood that the vessel will narrow again (restenosis) compared to standard balloons.
An EndoAVF is created using a catheter-based approach without open surgery. It uses magnets and radiofrequency energy to create a connection between the vein and artery. The advantage is that it avoids surgical incisions, preserves the natural surroundings of the vessels, leaves no visible scar, and may lead to fewer complications and better long-term flow rates compared to surgically created fistulas.
Kidney patients may require dialysis for many years, and each access site (fistula or graft) has a limited lifespan. Preserving veins involves avoiding unnecessary blood draws or IV lines in the arms to prevent scarring. This ensures that healthy veins are available for creating future dialysis access sites if the current one fails, essentially preserving the patient’s “lifeline.”
Yes, a blocked or thrombosed fistula can often be salvaged through interventional procedures. This typically involves a thrombectomy to remove the clot and an angioplasty to open the underlying narrowing (stenosis) that caused the clot to form. Prompt intervention, usually within 24 to 48 hours of the blockage, offers the best chance of restoring function and avoiding the need for a temporary catheter.

 Interventional Nephrology
Interventional Nephrology
Leave your phone number and our medical team will call you back to discuss your healthcare needs and answer all your questions.
Your Comparison List (you must select at least 2 packages)