Discover how the phosphate buffer system work maintains stable pH in the body, ensuring optimal enzyme function and metabolic reactions. Our bodies keep a fine chemical balance all the time. This is thanks to special buffering systems that stop big changes in acidity or alkalinity. Biological buffers are key chemical systems that help keep the body’s pH levels stable.
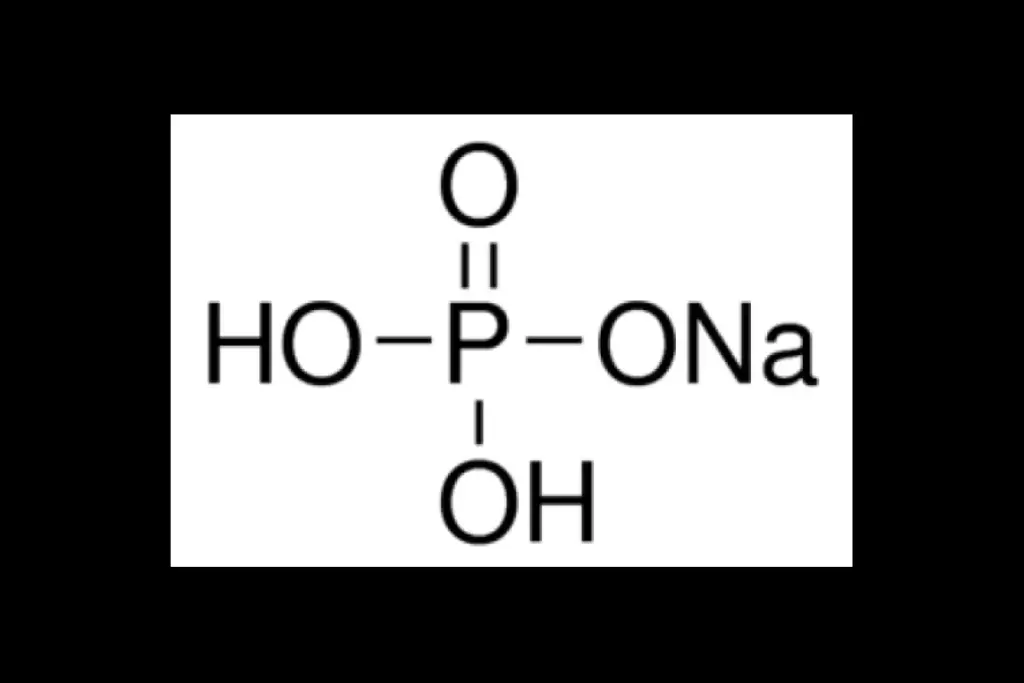
The phosphate buffer system is a big part of these buffering systems. It works mainly in the body’s cells. It uses dihydrogen phosphate ions (H2PO4-) and hydrogen phosphate ions (HPO42-) to fight off harmful acids and bases. For more info on biological buffers, check out this educational link.
Key Takeaways
- Biological buffers maintain stable pH levels in the human body.
- The phosphate buffer system operates mainly in intracellular fluids.
- Dihydrogen phosphate and hydrogen phosphate ions work together to neutralize acids and bases.
- Biological buffers are key for keeping the body’s acid-base balance.
- The phosphate buffer system is essential for the body’s functions.
Understanding Biological Buffers in the Human Body
Keeping the body’s pH in a tight range is vital. Biological buffers are essential for this. They help keep the acid-base balance right, which is key for the body to work well.
Definition and Importance of pH Regulation
Biological buffers are chemical systems that keep the body’s pH stable. They neutralize extra hydrogen or hydroxide ions. pH regulation is critical because many body processes are sensitive to pH changes. Even small pH shifts can cause big problems.
We need buffer systems to avoid these issues. They help enzymes and proteins work right. These are vital for metabolism and making energy.
Types of Biological Buffer Systems
The human body uses several buffer systems to keep acid-base balance. These include:
- The bicarbonate buffer system, key in blood plasma.
- The phosphate buffer system, important in cells and kidneys.
- Protein buffers, like hemoglobin, which can take or give hydrogen ions.
Each system has its own job. Together, they keep the body’s pH in a healthy range. Knowing about these systems helps us see how the body keeps its acid-base balance.
The Phosphate Buffer System: Mechanism and Function

The phosphate buffer system is key to keeping the pH inside cells stable. It’s important for many cell functions, like enzyme work and metabolic reactions.
Chemical Components: H2PO4- and HPO42-
This system has two main parts: dihydrogen phosphate (H2PO4) and hydrogen phosphate (HPO4) ions. These ions help keep pH levels steady by adding or removing hydrogen ions.
Key Components:
- Dihydrogen phosphate (H2PO4)
- Hydrogen phosphate (HPO4)
How Phosphate Buffers Neutralize pH Changes
When there’s too many hydrogen ions (H), the system gets acidic. Hydrogen phosphate ions (HPO4) then take these ions to form dihydrogen phosphate (H2PO4). On the other hand, when there’s not enough H ions, dihydrogen phosphate (H2PO4) gives up H ions to form hydrogen phosphate (HPO4).
Intracellular pH Maintenance (7.2-7.4)
The phosphate buffer system keeps the pH inside cells between 7.2 and 7.4. This range is vital for enzymes to work well and for metabolism to happen smoothly. Even small pH changes can affect enzyme activity a lot.
Importance for Enzyme Function and Metabolism
The phosphate buffer system’s role in keeping pH stable is critical for enzyme function and metabolism. Many enzymes are best suited to work in the pH range this system maintains.
| pH Range | Enzyme Activity | Metabolic Impact |
|---|---|---|
| 7.2-7.4 | Optimal | Normal metabolic processes |
| <7.2 | Reduced | Impaired metabolic processes |
| >7.4 | Altered | Potential metabolic dysfunctions |
Other Critical Buffer Systems in Human Physiology
Other than the phosphate buffer system, there are many other important buffer systems in our bodies. These systems work together to keep our acid-base balance right. This is key for our bodies to function properly.
The Bicarbonate Buffer System in Blood Plasma
The bicarbonate buffer system is a major player in blood plasma. It helps keep blood pH stable. It works with the respiratory and renal systems to adjust pH levels based on CO2 changes. This system uses bicarbonate ions and carbonic acid to neutralize extra hydrogen or hydroxide ions.
Key components of the bicarbonate buffer system include:
- Bicarbonate ions (HCO3)
- Carbonic acid (H2CO3)
- Carbon dioxide (CO2)
Protein Buffers and Their Role
Proteins, like hemoglobin, are important buffers. They can take in or give out hydrogen ions. This helps regulate pH levels in both inside and outside cells. Hemoglobin, for example, can release or bind hydrogen ions based on the pH around it.
Key aspects of protein buffers include:
- Ability to accept or donate hydrogen ions
- Presence in both intracellular and extracellular fluids
- Critical role of hemoglobin in buffering
Timeframes for Buffer Responses
Buffer systems in our bodies react to pH changes at different speeds. The bicarbonate buffer system, for example, can quickly adjust to CO2 changes through breathing. But, the kidneys take longer to adjust pH levels by changing how much bicarbonate they reabsorb and hydrogen ions they excrete.
Timeframes for buffer responses vary:
- Immediate responses: Chemical buffers like bicarbonate and proteins
- Rapid responses: Respiratory adjustments to CO2 levels
- Slow responses: Renal adjustments to bicarbonate and hydrogen ion levels
Conclusion: The Vital Role of Buffer Systems in Maintaining Homeostasis
Buffer systems are key to keeping our body’s acid-base balance right. They make sure our body’s processes work well. A biological buffer is a solution that doesn’t change pH much, even when acids or bases are added.
We’ve seen how different buffers, like the phosphate and bicarbonate systems, help keep us balanced. Knowing about biological buffers helps us understand how our body stays in balance.
For example, the blood’s carbonic acid and bicarbonate system keeps blood pH safe. By learning about biological buffers, we see how they help our body work right.
In short, buffers are vital for keeping our body’s pH levels stable. This lets our body’s processes work as they should. By knowing about buffers, we can see how they keep us healthy.
FAQ
What are biological buffers?
Biological buffers are chemical systems. They help keep the body’s acid-base balance right. They do this by controlling pH levels in different bodily fluids.
What is the phosphate buffer system, and how does it work?
The phosphate buffer system is key in keeping the body’s pH right. It works mainly in the cells. It does this by taking or giving hydrogen ions.
Why is the phosphate buffer system important in the human body?
The phosphate buffer system is vital for keeping the pH of cell fluids right. It’s important in the kidneys. It helps enzymes work and supports metabolic processes.
What are the different types of buffer systems in the human body?
The human body has several buffer systems. These include the phosphate, bicarbonate, and protein buffers. They all work together to keep the pH stable.
How does the bicarbonate buffer system work?
The bicarbonate buffer system is key in blood plasma. It helps control pH by adjusting to CO2 changes.
What is the role of protein buffers in the human body?
Protein buffers, like hemoglobin, help with pH control. They do this in both cell and body fluids. This is because of their ionizable side chains.
What is the significance of maintaining optimal pH levels in the body?
Keeping pH levels right is key for the body’s functions. Many enzymes need a specific pH to work well.
How do buffer systems work together to maintain acid-base balance?
The phosphate, bicarbonate, and protein buffers work together. They make sure the body’s pH stays within a healthy range.
Reference
https://pmc.ncbi.nlm.nih.gov/articles/PMC8956001




































